Knowing the Unknowable: How We Discovered What Stars Are Made Of
There is more to starlight than meets the eye.
Are there limits to what science can discover? Philosopher of science Auguste Comte certainly thought so. Indeed, he had a simple example to prove it. We can never know what stars are made of.1 The reasoning was simple. We can never travel to the stars, since they are too far away and too hot. Therefore, we can never obtain a sample of a star to analyse in the laboratory. So, we can never determine what they are made of.
Writing in 1835, this conclusion is understandable. Indeed, it must have seemed vindicated by the history of progress on the question to that date. Astronomy may be the oldest of all the sciences. Humans have been astronomers since before they could write. From Britain to Egypt to Peru, ancient peoples all around the world went to incredible efforts to construct observatories from which they could study the skies. From ancient Babylonian astrologers to the greatest minds of the Scientific Revolution, people had studied the stars in exquisite detail for millennia in an effort to uncover the deepest truths of the world. Yet for all this attention on the stars, little more could be said about their nature than that they were probably distant and hot. What reason could there be to think this would ever change?

Yet, in fact, Comte was very wrong! Within a generation of Comte’s writing, the new science of spectroscopy was already unravelling the mysteries of the stars. Far from being forever unknowable, today we can describe the composition of stars in exquisite detail. Indeed, it turns out all the evidence we needed was right in front of our eyes every night. We just needed to learn how to see it.
A Spectrum of Possibilities
To the naked eye, there is not much to see in starlight. All stars look like more or less white points in the night sky, with relatively little to distinguish them. Yet, hidden in every ray of starlight is an incredibly detailled biography of the star that produced it. A sufficiently knowledgeable observer with the right tools can use a star’s light to determine its composition, temperature, age, size, distance from Earth and motion.
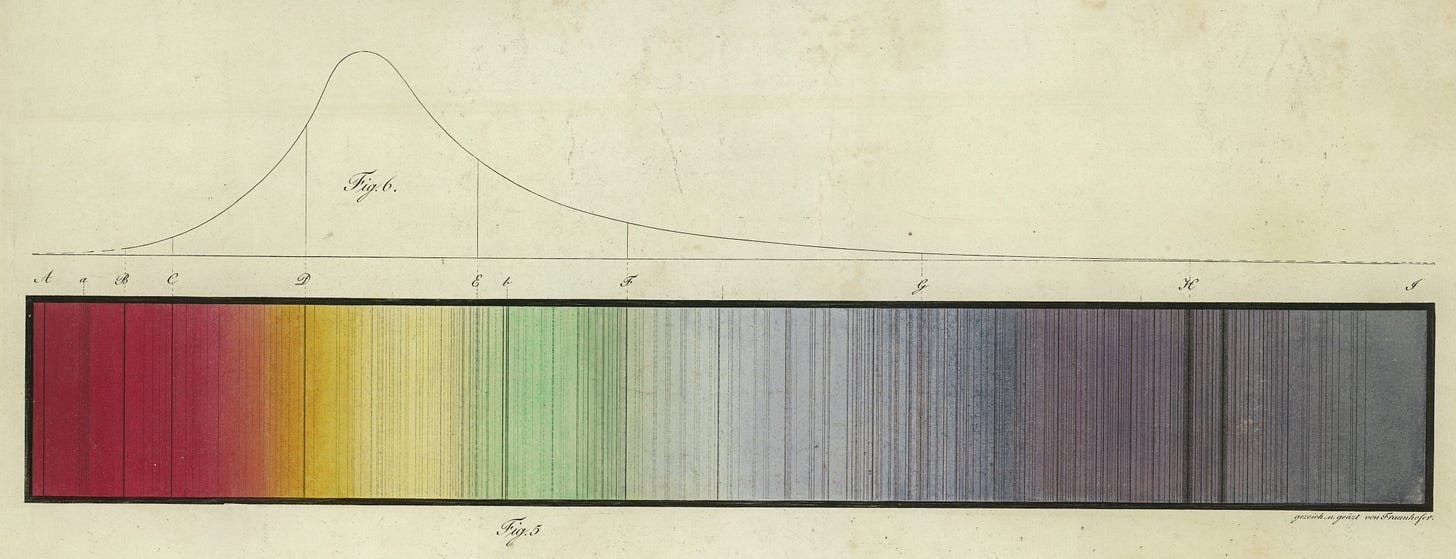
The trick to uncovering this information lies in splitting starlight into its constituent colours. The first person to do this was Isaac Newton. He discovered that when sunlight is passed through a glass prism, it splits into a spectrum containing all of the colours of the rainbow. From this, he concluded that the white light of the Sun (and other stars) is formed out of the combination of all of these colours. In 1814, Joseph von Fraunhofer refined Newton’s prism into a spectroscope. This allowed far more fine detail of the spectra of the Sun and stars to be observed. In particular, Fraunhofer discovered that scattered throughout the light of the spectrum were a series of thin black lines, corresponding to colours that were missing from the star’s light. These came to be called spectral lines. Fraunhofer didn’t realise it, but these lines held the key to uncovering the mysteries of stars.
To understand why, we must consider how absorption of light by matter can occur. Light is made up of tiny particles called photons. Each photon contains a certain amount of energy. This energy is determined by the colour of the light; for visible light, red photons contain the lowest energy, followed by orange, then yellow, then green, then blue, then indigo, then violet.2 Absorption of light occurs when a photon transfers all of its energy to an atom in matter, and is thereby destroyed.
Specifically, the photons can be absorbed by particles within atoms called electrons. Electrons in an atom can exist in a number of different states, corresponding to different levels of energy. We can think of each electron as analogous to a ball sitting on a staircase; the ball can sit on any one of the steps and has more energy the higher up the staircase it is. If a photon is absorbed by an electron, the additional energy transferred to the electron can move the electron up to a higher energy level. However, absorption can only occur if the photon has just the right amount of energy to move the electron exactly into another energy level. In the analogy, we can think of the photon as kicking the ball up the staircase; if the ball lands on another step then it can stay there, but otherwise it falls back down to its original step and gives all the extra energy back to what kicked it.
This means that when white light passes through a gas, only certain energies of photons (and so, only certain colours of light) will be absorbed. This means that if you look at the light that passes out the other side through a spectroscope, you will see a spectrum of colours punctuated by a pattern of dark lines wherever there is a colour that was absorbed. This is just what Fraunhofer observed! Indeed, the reason for the lines in the spectrum of a star (such as the Sun) is that the light of a star is produced in its core. To reach our eyes, it must escape from the centre of the star to travel across space to Earth. The particular colours that are absorbed by the gas in the star will therefore not reach us, resulting in the spectral lines.
Star Power
Spectral lines allow for the mystery of the composition of stars to be solved. This is because every element of the periodic table has a unique set of spectral lines. So, the lines in a stellar spectrum can be matched to those observed for different elements in the laboratory to identify which elements must be present in the star. Moreover, comparing how dark different lines appear can allow for the relative proportions of their corresponding elements in the star to be inferred. In this way, the chemical composition of stars can be determined without any recourse to interstellar travel.
So, what are stars? In 1925, Cecilia Payne-Gapsochkin worked through the analysis to finally publish the answer. They are mostly hydrogen with some helium and traces of other elements. Mystery solved! Isn't that satisfying? Unless you are blessed with an unusual passion for relative abundances of gases, then, alas, perhaps not. Like Loonquawl and Phouchg hearing the answer 42 in The Hitchhiker's Guide to the Galaxy, we may well wonder whether this is really the answer to the right question. When, in 1806, Jane Taylor wrote “Twinkle, twinkle little star, how I wonder what you are”, her hope was surely for a deeper answer than “73% hydrogen and 25% helium”.
Fortunately, this is not the end of the story. There are genuine insights in this ostensibly dry data. To begin with, it confirms that stars are fundamentally made of the same type of matter we find on Earth. There is no magic ingredient that powers the stars; they truly are nothing but extremely hot balls of gas. Yet this leaves the question: what does power the stars? As discussed in Oxygen: The Killer We Can’t Live Without, conventional burning describes reacting fuel with oxygen. But we now know that oxygen is largely absent from stars. So, stars cannot simply be giant balls of fire. What alternative could there be?
Fortunately, the composition of stars provides the crucial clue. Since stars are almost entirely made of hydrogen and helium, we are looking for a process that involves only these elements. We know of one such process — nuclear fusion. This involves fusing together hydrogen atoms into larger helium atoms in a reaction that releases immense quantities of energy. For decades, scientists have worked tenaciously to emulate this power source on Earth to create a safe, clean and virtually unlimited source of energy. Unfortunately, they have so far been stymied by the challenge of reproducing the temperatures of the core of the Sun inside a power plant, although recent progress has been promising. In the meantime, we can appreciate that, through our study of the stars, we now understand the process that powers the Earth and all of its life. Hopefully that, at least, is a satisfying conclusion.
Comte’s Mistake
With our quest to understand the composition of stars now complete, let us return to the man who declared it impossible. How could Auguste Comte have been so wrong? At a general level, Comte’s mistake was simply overconfidence. He mistook the unimaginable for the impossible. In this error, he is certainly not alone. He at least took longer to be proven wrong than the great physicist Lord Kelvin who declared that “heavier than air flying machines are impossible” just eight years before they were invented. Perhaps we must simply learn to take such pronouncements with a grain of salt.
Yet in the case of Comte, there is also a more specific lesson. Science does not only progress by the discovery of new evidence; it also advances by finding new ways to look at old evidence. In his statement, Comte specifically emphasised that “on the subject of stars, all investigations which are not ultimately reducible to simple visual observations” are impossible. He took it as self-evident that simple visual observations could not allow us to determine the composition of stars. Comte understood that starlight is there to observe and that we can learn about the stars from it, but he erred in believing that visual observations of it must be “simple”. Stellar spectroscopy is nothing more than a sophisticated way of looking at stars. Yet that innovation revealed that the same starlight that humanity had witnessed for millennia still contained secrets that had seemed forever beyond reach. Ultimately, the basic fact that undid Comte’s prediction is simple: There is more to starlight than meets the eye.
Specifically, he wrote: “On the subject of stars, all investigations which are not ultimately reducible to simple visual observations are… necessarily denied to us… We shall never be able by any means to study their chemical composition.”
The colour of light also corresponds to its frequency. Max Planck showed that the energy of a photon is proportional to its frequency through his equation E=hf.


Spectroscopy is so cool :) One question about it: Is it possible to construct two samples made of different elements which have an indistinguishable spectrum? Or are the spectral 'fingerprints' of each element sufficiently unique?
Sounds like a problem that one could formalize in linear algebra, maybe ... if we treat each wavelength as a dimension in a real-valued vector space and each spectral fingerprint as a vector, the question is whether the set of all these vectors is linearly independent, I think.
Another article explaining stuff so well! I assume there is some limit to spectroscopy’s ability to detect a multiple number of different elements in that once you get above a certain complexity of elements there would be overlaps (like overpaying multiple QR codes) in the spectroscopic image and thus some degree of ambiguity could set in. Or maybe this is resolved through obtaining finer and finer images